Specimen Interaction
 Specimen interaction is what
makes Electron Microscopy possible. The energetic electrons in
the microscope strike the sample and various reactions can occur
as shown below. The reactions noted on the top side of the diagram
are utilized when examining thick or bulk specimens(SEM) while
the reactions on the bottom side are those examined in thin or
foil specimens (TEM). A diagram showing the generation depths
of the interactions is also available
Specimen interaction is what
makes Electron Microscopy possible. The energetic electrons in
the microscope strike the sample and various reactions can occur
as shown below. The reactions noted on the top side of the diagram
are utilized when examining thick or bulk specimens(SEM) while
the reactions on the bottom side are those examined in thin or
foil specimens (TEM). A diagram showing the generation depths
of the interactions is also available
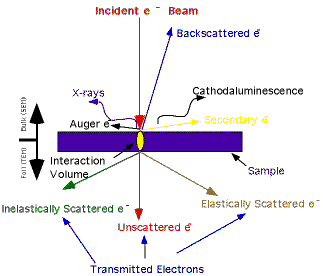
The formation and uses of these reactions:
Bulk Specimen Interactions
- Backscattered Electrons:
-
- Formation
- Caused by an incident electron colliding with an atom in the
specimen which is nearly normal to the incident's path. The incident
electron is then scattered "backward" 180 degrees.
- Utilization
- The production of backscattered electrons varies directly
with the specimen's atomic number. This differing production rates
causes higher atomic number elements to appear brighter than lower
atomic number elements. This interaction is utilized to differentiate
parts of the specimen that have different average atomic number.
(See Interaction Volume) An example is
shown in the SEM output section, specifically the mechanically
alloyed specimen micrograph.
- Secondary Electrons:
-
- Source
- Caused by an incident electron passing "near" an
atom in the specimen, near enough to impart some of its energy
to a lower energy electron (usually in the K-shell). This causes
a slight energy loss and path change in the incident electron
and the ionization of the electron in the specimen atom. This
ionized electron then leaves the atom with a very small kinetic
energy (5eV) and is then termed a "secondary electron".
Each incident electron can produce several secondary electrons.
- Utilization
- Production of secondary electrons is very topography related.
Due to their low energy, 5eV, only secondaries that are very near
the surface (<10nm, seeInteraction Volume)
can exit the sample and be examined. Any changes in topography
in the sample that are larger than this sampling depth will change
the yield of secondaries due to collection efficiencies. Collection
of these electrons is aided by using a "collector" in
conjunction with the secondary electron detector. The collector
is a grid or mesh with a +100V potential applied to it which is
placed in front of the detector, attracting the negatively charged
secondary electrons to it which then pass through the grid-holes
and into the detector to be counted.
- Auger Electrons
-
- Source
- Caused by the de-energization of the specimen atom after a
secondary electron is produced. Since a lower (usually K-shell)
electron was emitted from the atom during the secondary electron
process an inner (lower energy) shell now has a vacancy. A higher
energy electron from the same atom can "fall" to a lower
energy, filling the vacancy. This creates and energy surplus in
the atom which can be corrected by emitting an outer (lower energy)
electron; an Auger Electron.
- Utilization
- Auger Electrons have a characteristic energy, unique to each
element from which it was emitted from. These electrons are collected
and sorted according to energy to give compositional information
about the specimen. Since Auger Electrons have relatively low
energy they are only emitted from the bulk specimen from a depth
of <3see Interaction Volume).
- X-rays
-
- Source
- Caused by the de-energization of the specimen atom after a
secondary electron is produced. Since a lower (usually K-shell)
electron was emitted from the atom during the secondary electron
process an inner (lower energy) shell now has a vacancy. A higher
energy electron can "fall" into the lower energy shell,
filling the vacancy. As the electron "falls" it emits
energy, usually X-rays to balance the total energy of the atom
so it .
- Utilization
- X-rays or Light emitted from the atom will have a characteristic
energy which is unique to the element from which it originated.
These signals are collected and sorted according to energy to
yield micrometer
diameter) of bulk specimens limiting the point-to-point comparisons
available (see Interaction Volume).
Thin Specimen Interactions
- Unscattered Electrons
-
- Source
- Incident electrons which are transmitted through the thin
specimen without any interaction occurring inside the specimen.
- Utilization
- The transmission of unscattered electrons is inversely proportional
to the specimen thickness. Areas of the specimen that are thicker
will have fewer transmitted unscattered electrons and so will
appear darker, conversely the thinner areas will have more transmitted
and thus will appear lighter.
- Elasticity Scattered electrons
-
- Source
- Incident electrons that are scattered (deflected from their
original path) by atoms in the specimen in an elastic fashion
(no loss of energy). These scattered electrons are then transmitted
through the remaining portions of the specimen.
- Utilization
- All electrons follow Bragg's Law and thus are scattered according
to Wavelength=2*Space between the atoms in the specimen*sin(angle
of scattering). All incident electrons have the same energy(thus
wavelength) and enter the specimen normal to its surface. All
incidents that are scattered by the same atomic spacing will be
scattered by the same angle. These "similar angle" scattered
electrons can be collated using magnetic lenses to form a pattern
of spots; each spot corresponding to a specific atomic spacing
(a plane). This pattern can then yield information about the orientation,
atomic arrangements and phases present in the area being examined.
- Inelastically Scattered Electrons
-
- Source
- Incident electrons that interact with specimen atoms in a
inelastic fashion, loosing energy during the interaction. These
electrons are then transmitted trough the rest of the specimen
- Utilization
- Inelasticaly scattered electrons can be utilized two ways
- Electron Energy Loss Spectroscopy: The inelastic loss of energy
by the incident electrons is characteristic of the elements that
were interacted with. These energies are unique to each bonding
state of each element and thus can be used to extract both compositional
and bonding (i.e. oxidation state) information on the specimen
region being examined.
- Kakuchi Bands: Bands of alternating light and dark lines that
are formed by inelastic scattering interactions that are related
to atomic spacings in the specimen. These bands can be either
measured (their width is inversely proportional to atomic spacing)
or "followed" like a roadmap to the "real"
elasticity scattered electron pattern.
![]() Specimen interaction is what
makes Electron Microscopy possible. The energetic electrons in
the microscope strike the sample and various reactions can occur
as shown below. The reactions noted on the top side of the diagram
are utilized when examining thick or bulk specimens(SEM) while
the reactions on the bottom side are those examined in thin or
foil specimens (TEM). A diagram showing the generation depths
of the interactions is also available
Specimen interaction is what
makes Electron Microscopy possible. The energetic electrons in
the microscope strike the sample and various reactions can occur
as shown below. The reactions noted on the top side of the diagram
are utilized when examining thick or bulk specimens(SEM) while
the reactions on the bottom side are those examined in thin or
foil specimens (TEM). A diagram showing the generation depths
of the interactions is also available
