
Professor of Microbiology and Immunology
Dept. Oral Biology
University of Nebraska Medical Center
College of Dentistry
40th and Holdrege
Lincoln, NE 68583-0740
402-472-1327
Fax: 402-472-2551
tpetro@unmc.edu
Research Interest
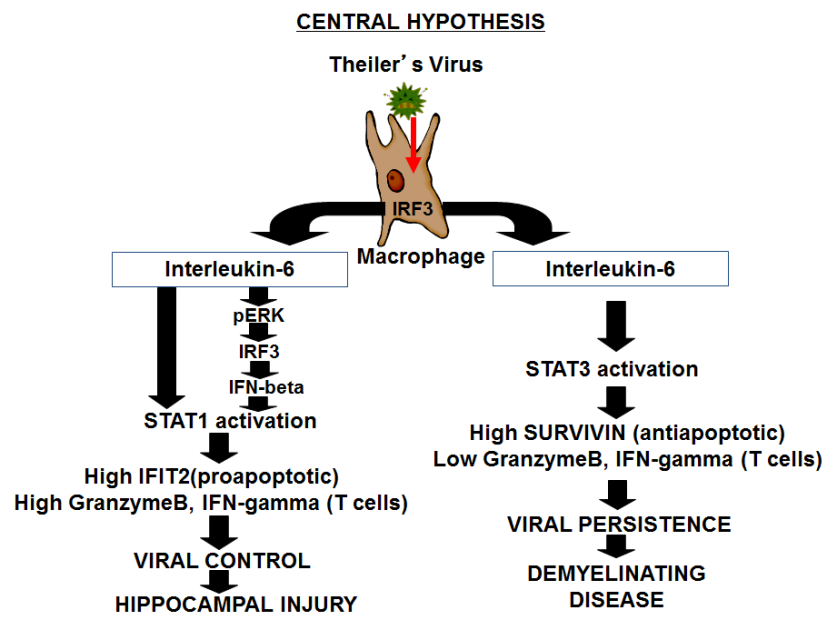
All humans are infected with viruses throughout their lifetime. While most of these virus infections stimulate innate and adaptive immunity and are eliminated, it is estimated that each human is infected by 8-10 viruses that persistently stimulate the immune systems and are never eliminated. It is hypothesized that in some cases these persistent viral infections lead to diseases, such as Multiple Sclerosis (MS). As a model for persistent viral infections, his laboratory is focusing upon the innate immune response to the Theiler's Murine Encephalomyelitis Virus (TMEV). In some strains of mice, TMEV persistently infects macrophages of the immune system but is never eliminated within the lifetime of the individual mouse. Those mice strains end up with demyelination and symptoms like MS. In other strains of mice, TMEV is eliminated within a few weeks. However, the immune response to eliminate the virus is not without consequence and these mice end up with damage to the hippocampus region of the brain. We showed that failures within the innate immune system are causes of TMEV persistence but also protect the mice from immune mediated damage to host tissues. The innate immune response to viral infection depends on Interferon Response Factor-3 (IRF-3), which plays a key role in induction of Type I interferons (IFN-alpha/beta), IL-6, and IL-12. We showed that IL-6 induces activation of STAT1 and ERK and thus is a key antiviral cytokine during this response to TMEV. We hypothesize that robust activation of IRF3 induces a high level of IL-6 expression that contributes to viral control but also causes damage to the hippocampus. We have shown that functional IRF3 is related to viral clearance but is also related to hippocampal damage. In addition to innate cytokine production, IRF-3 is also involved in the induction of apoptosis (programmed cell death) that is triggered in virus-infected cells. Apoptosis prevents viral persistence and infection into surrounding cells. We have also found that IRF-3 of mice that cannot eliminate TMEV differs from IRF-3 of mice that can control this virus, in that it cannot participate in virus induced apoptosis as well. Therefore our central hypothesis is that differences in host IRF-3 underlie persistent infections with certain viruses but also immune mediate damage during viral infections.
Selected Abstracts
- Petro, TM and Zhang, Y, Theiler's virus induction of p35, p40 and p19 IL-12 family subunits is reduced by a p38 MAP-kinase inhibitor.Presented at the American Association of Immunology Annual Meeting, Denver, CO, May 2003
- Kollet, J, and Petro, TM. In vivo and in vitro interaction of IRF-1 and c-Rel with the proximal Interleukin p35 promtoer. Presented at the American Association of Immunology Annual Meeting, Denver, CO, May 2003
- Kollet J, Witek C, Gentry JD, Liu X, Schwartzbach SD, and Petro TM. Recruitment of Interferon Regulatory Factor 1 (IRF-1) to the Interleukin-12 recruitment of Interferon Regulatory Factor 1 (IRF-1) to the Interleukin-12 p35 Promoter upon Interferon-[gamma]/LPS Stimulation35 Promoter upon Interferon-_/LPS Stimulation. 41st American Society for Cell Biology Annual Meeting in Washington, DC, December 8-12, 2001.
- Petro, T.M., Witek, C., Kollet, J., Liu, X., Schwartzbach, S.D. Distinct regulatory elements in the murine IL-12 p35 promoter respond to Interferon-_ and LPS. Presented at the American Association of Immunology Annual Meeting, Seattle, WA, FASEB J 14(6):A1182:#155, April 14, 2000.
- Petro, T.M., Gentry, J.D., Weatherman, A., Schwartzbach, S.D. Analysis of murine IL-12 p 35 promoter. Experimental Biology, 1999, Washington, D.C., FASEB J 13(5):A651, March 15, 1999.
Selected Research Publications
- Moore TC, Petro TM. IRF3 and ERK MAP-kinases control nitric oxide production from macrophages in response to poly-I:C. FEBS Lett. 587(18):3014-20, 2013.
- Moore TC, Cody L, Kumm PM, Brown DM, Petro TM. IRF3 helps control acute TMEV infection through IL-6 expression but contributes to acute hippocampus damage following TMEV infection. Virus Res. 178(2):226-33, 2013.
- Moore TC, Bush KL, Cody L, Brown DM, Petro TM, Control of Early Theiler's Murine Encephalomyelitis Virus Replication in Macrophages by Interleukin-6 Occurs in Conjunction with STAT1 Activation and Nitric Oxide Production. J Virol. 86(19):10841-51, 2012.
- Moore TC, Al-Salleeh FM, Brown DM, and Petro TM, IRF3 Polymorphisms Induce Different Innate Anti-Theiler’s Virus Immune Responses in RAW264.7 Macrophages. Virology 15;418(1):40-8, 2011.
- Petro TM, Regulatory role of resveratrol on Th17 in autoimmune disease, Int Immunopharmacol.11(3):310-8, 2011.
- Ghosh, S, Howe, N, Volk, K, Tati, S. Nickerson, KW, Petro, TM. Candida albicans Through Beta-glucan and Farnesol Stimulates Expression of Th17-Inducing, Inflammatory, and Regulatory Cytokines in the Murine RAW264.7 Macrophage Cell Line. FEMS Immunol Med Microbiol. 60: 63-73, 2010.
- Ghosh S, Navarathna DH, Roberts DD, Cooper JT, Atkin AL, Petro TM, Nickerson KW. Arginine-induced germ tube formation in Candida albicans is essential for escape from murine macrophage line RAW 264.7. Infect Immun. 77(4):1596-605, 2009.
- Imler TJ and Petro TM. Decreased Severity of Experimental Autoimmune Encephalomyelitis During Resveratrol Administration is Associated with Increased IL-17+IL10+ T cells, CD4- IFN-g+ cells, and Decreased Macrophage IL-6 Expression. International Immunopharmacology. 9(1):134-43. 2009.
- Al-Salleeh FM and Petro TM. Promoter analysis reveals critical roles for IRF-3, SMAD-3 and ATF-2 in expression of IL-23 p19 in RAW264.7 cells. J. Immunol. 181: 4523-4533, 2008.
- Al-Salleeh F, Beatty MW, Reinhardt RA, Petro TM, Crouch L. Human osteogenic protein-1 induces osteogenic differentiation of adipose-derived stem cells harvested from mice. Arch Oral Biol. 53: 928-936 2008.
- Al-Salleeh FM and Petro TM. TLR3 and TLR7 are involved in expression of IL-23 subunits while TLR3 but not TLR7 is involved in expression of IFN- by Theiler’s virus-infected RAW264.7 cells. Microbes and Infection. 11:1384-92, 2007.
- Hause L, Al-Salleeh FM and Petro TM. Expression of IL-27 p28 by Theiler’s virus-Infected Macrophages Depends on TLR3 and TLR7 Activation of JNK-MAP-Kinases. Antiviral Research. 76(2) 159-167, 2007.
- Navarathna DH, Nickerson KW, Duhamel GE, Jerrels TR, Petro TM. Exogenous farnesol interferes with the normal progression of cytokine expression during candidiasis in a mouse model. Infect Immun. 75(8):4006-11, 2007.
- Kollet, JI and Petro TM. IRF-1 and NF-B p50/cRel Bind to Distinct Regions of the Proximal Murine IL-12 p35 Promoter During Costimulation with IFN-g and LPS. Molecular Immunology 43: 623-633, 2006.
- Dahlberg, A, Auble, MR, and Petro TM. Reduced expression of IL-12 p35 by SJL/J macrophages responding to Theiler’s virus infection is associated with constitutive activation of IRF-3. Virology 353: 422-432, 2006.
- Petro, TM. ERK-MAP-kinases differentially regulate expression of IL-23 p19 compared with p40 and IFN-beta in Theiler's virus-infected RAW264.7 cells. Immunol Lett. 15;97(1):47-53, 2005.
- Petro, TM. Disparate expression of IL-12 by SJL/J and B10.S macrophages during Theiler's virus infection is associated with activity of TLR7 and Mitogen-activated Protein Kinases, Microbes and Infection 7: 224-232, 2005.
- Vaidyanathan H, Zhou Y, Petro TM, and Schwartzbach SD. Intracellular localization of the p35 subunit of murine IL-12. Cytokine, 21:120-128, 2003.-
Education
1977 - 1980 PhD Immunology/Microbiology, Miami University, Oxford, Ohio
1975 - 1977 MS Microbiology, Miami University, Oxford, Ohio
1972 - 1975 BS Microbiology, Ohio State University, Columbus, Ohio
Professional Positions
July 2002 - Present Professor, Department of Oral Biology, University of Nebraska Medical Center, College of Dentistry, Lincoln, Nebraska
1992 - Present Courtesy Appointment, Associate Professor, Dept. of Veterinary & Biomedical Science, University of Nebraska-Lincoln
1991 - 2002 Associate Professor
Department of Oral Biology
University of Nebraska Medical Center
College of Dentistry
Lincoln, NE
1990 - Present Faculty Associate, UNL Center for Biotechnology
1987 - 1991 Assistant Professor, Department of Oral Biology, University of Nebraska Medical Center, College of Dentistry, Lincoln, NE 68583
1984 - 1987 Microbiologist, Microbial Biochemistry Branch, Division of Microbiology, Food and Drug Administration, Cincinnati, OH 45226
1982 - 1984 Visiting Assistant Professor, Department of Foods & Nutrition and Department of Biological Sciences, Purdue University, West Lafayette, IN 47907
1980 - 1982 Postdoctoral Associate (Preceptor Ronald R. Watson, PhD)
Immunology/Nutrition
Purdue University
West Lafayette
Indiana